How Does Ozone Layer Depletion Affect Life on Earth for NEET Students?
Ozone Layer and Its Depletion is a vital topic in Biology for NEET aspirants, as it connects atmospheric science to environmental biology and human health. Understanding what the ozone layer is, how it gets depleted, its causes and consequences, and related sub-concepts not only helps in direct questions but also in grasping wider biological impacts of pollution. This page explains the concept clearly and offers practical guidance for NEET preparation, along with images and revision tips for thorough understanding.
What is the Ozone Layer and Its Depletion?
The ozone layer is a protective layer of gas located in the Earth’s stratosphere, roughly 10 to 50 km above the surface. It is composed mainly of ozone (O3) molecules, which absorb the majority of the sun’s harmful ultraviolet (UV) radiation. Ozone layer depletion refers to the thinning or reduction of this protective layer, mainly due to human-made chemicals like chlorofluorocarbons (CFCs), halons, and others. The depletion of the ozone layer allows more UV rays to reach Earth's surface, leading to serious environmental and health effects—which make it an essential concept in the NEET Biology syllabus.
Core Ideas of Ozone Layer and Its Depletion
Location and Structure of the Ozone Layer
The ozone layer is found in the stratosphere, around 10 to 50 km above Earth. It is not a thick blanket but a region where ozone concentration is higher compared to other parts of the atmosphere.
Formation of Ozone
Ozone forms when ultraviolet (UV) light from the sun splits oxygen molecules (O2) into individual oxygen atoms. These atoms combine with other O2 molecules to form ozone (O3).
Role of the Ozone Layer
The primary function of the ozone layer is to absorb most of the sun’s harmful UV-B and UV-C radiation, preventing it from reaching living organisms on Earth. Without this layer, life as we know it would not be possible due to increased rates of diseases and ecological disturbance.
Ozone Layer Depletion: Causes and Consequences
Ozone depletion occurs mainly due to human-made chemicals such as CFCs, halons, and certain solvents. These chemicals release chlorine and bromine atoms in the stratosphere, which react with ozone molecules and break them down. The most well-known example of depletion is the 'ozone hole' over Antarctica reported since the 1980s.
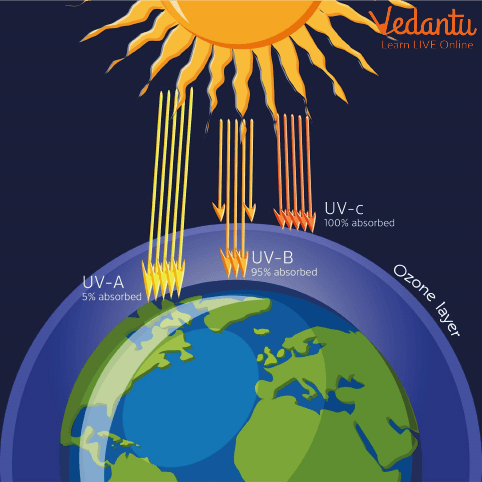
The above diagram illustrates how ozone layer depletion happens in the atmosphere.
Important Sub-Concepts Related to Ozone Layer and Its Depletion
1. Ultraviolet (UV) Radiation
UV radiation consists of high-energy rays from the sun, which can cause skin cancer, cataracts, and harmful mutations in DNA when not properly absorbed by the ozone layer. This highlights why ozone depletion is so dangerous for living beings.
2. Ozone-Depleting Substances (ODS)
ODS are chemicals like CFCs, halons, and carbon tetrachloride that erode the ozone layer. They are commonly used in refrigeration, air conditioning, foam-blowing agents, and aerosol propellants. Understanding ODS is essential to grasping the human impact on atmospheric processes.
3. The Ozone Hole
The ozone hole refers to the severe thinning of the ozone layer, particularly over Antarctica, observed during the Southern Hemisphere’s spring. It is a major example studied for environmental damage due to human activities.
4. Montreal Protocol
The Montreal Protocol is an international agreement signed in 1987 to phase out the production and use of ODS globally. Its success is a positive example of global cooperation for environmental protection, often mentioned in NEET-related discussions.
Key Principles and Relationships in Ozone Layer and Its Depletion
Some important scientific relationships and processes you should remember as a NEET aspirant:
- Ozone Formation: O2 + UV light → 2O; O + O2 → O3
- Ozone Destruction by CFCs: Cl from CFC reacts with O3 (Ozone), leading to the breakdown into O2.
- Each chlorine atom from CFCs can destroy thousands of ozone molecules before being neutralized.
- Presence of polar stratospheric clouds in Antarctica enhances the reaction rate leading to rapid ozone depletion ('ozone hole').
Effects of Ozone Layer Depletion
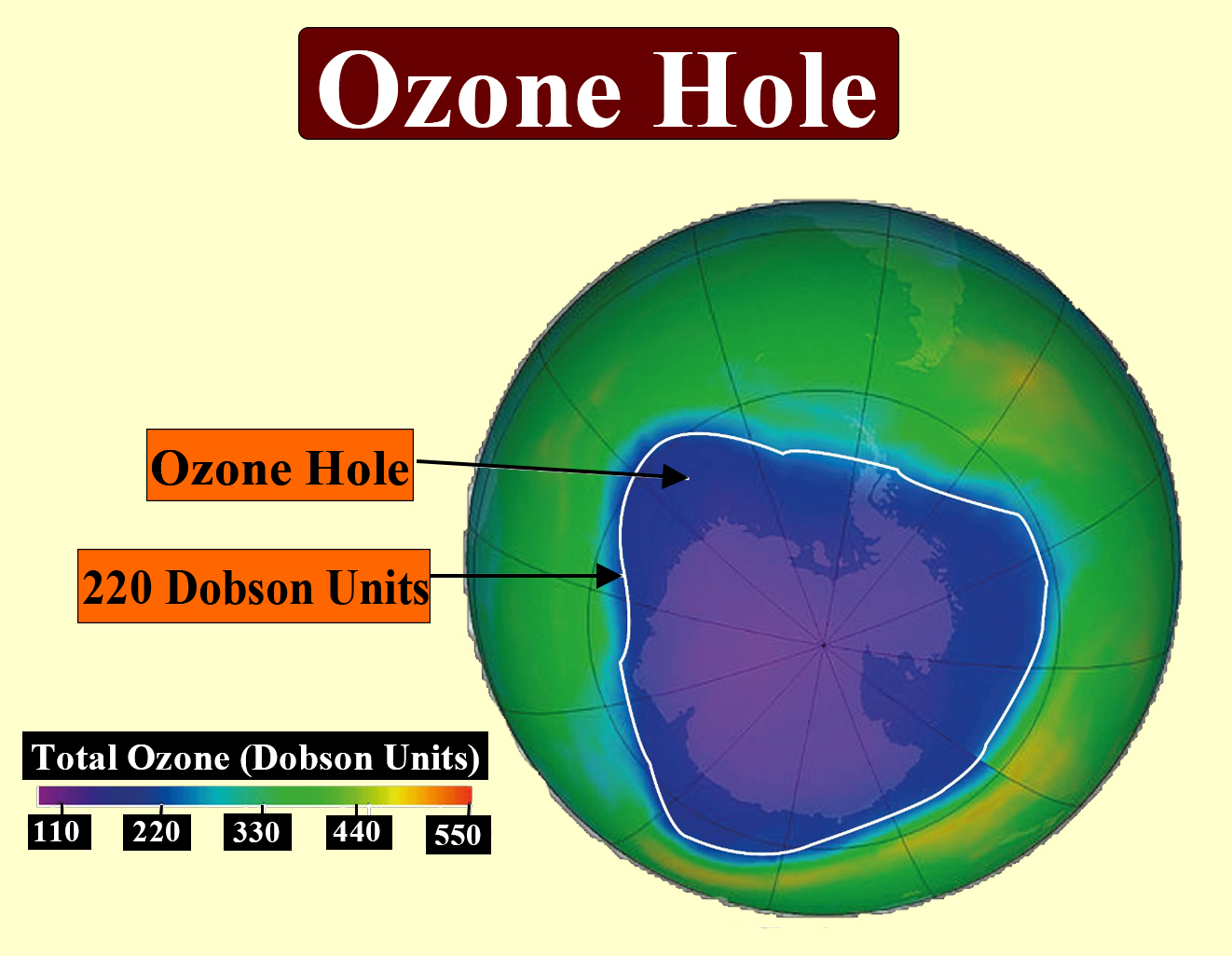
- Increased skin cancer risk due to higher UV exposure
- Higher chances of cataracts and eye damage
- Suppression of immune system functions
- Reduced crop productivity and negative effects on phytoplankton, impacting food chains
- Material degradation (e.g., plastics, paints) due to UV radiation
The above image and list summarize the main effects of ozone layer depletion, which are frequently discussed in NEET questions.
Importance of Ozone Layer and Its Depletion for NEET
This concept is often tested in NEET for its blend of environmental biology, atmospheric chemistry, and impact on health. Understanding ozone and its depletion helps students answer questions in Ecology & Environment, and even Genetics (mutations due to UV). It also helps build awareness about human-induced environmental issues, a theme that recurs across the Biology syllabus.
How to Study Ozone Layer and Its Depletion Effectively for NEET
- Read NCERT lines carefully, as NEET often frames its questions directly from textbooks.
- Understand processes like ozone formation, depletion, and cycling rather than memorizing definitions.
- Go through diagrams (such as the ones above) and practice labeling them for visual memory.
- Learn the list of ozone-depleting substances and their applications.
- Revise environmental treaties like the Montreal Protocol and their significance.
- Practice previous years’ MCQs and assertion-reason type questions based on this topic.
- Summarize the consequences of ozone depletion and learn to logically explain them in short.
Common Mistakes Students Make in This Concept
- Confusing between the ozone layer and greenhouse gases – both are important, but have very different functions.
- Misidentifying ODS or forgetting examples like CFCs, halons, methyl bromide, etc.
- Ignoring the difference between ozone depletion and global warming/climate change.
- Not revising environmental treaties, which are frequently asked as factual MCQs.
- Overlooking diagram-based questions or not practicing visual interpretation.
Quick Revision Points for Ozone Layer and Its Depletion
- Ozone (O3) protects life by blocking UV radiation.
- Ozone layer is located in the stratosphere (10-50 km above Earth).
- Main causes of depletion: CFCs, halons, and other ozone-depleting substances.
- Ozone hole is most evident over Antarctica.
- Health impacts: skin cancer, eye damage, weakened immune system.
- Montreal Protocol is the key treaty to stop ozone depletion.
- Learn to identify and label ozone layer depletion diagrams.
- Never confuse ozone depletion with global warming – they are separate phenomena.
FAQs on Ozone Layer And Its Depletion for NEET Biology
1. What is the ozone layer and why is it important for NEET Biology?
The ozone layer is a concentrated region of ozone (O3) molecules in the upper stratosphere that protects life on Earth by absorbing most of the Sun's harmful ultraviolet (UV) radiation. Its importance for NEET includes:
- Filters UV-B and UV-C rays that cause skin cancer and eye damage
- Maintains climatic balance and supports ecosystems
- Prevents harmful mutations in living organisms
- Acts as a key topic for NEET exam biology syllabus
2. What are the main causes of ozone layer depletion?
Ozone layer depletion is primarily caused by human-made chemicals called ozone-depleting substances (ODS). The main causes include:
- Chlorofluorocarbons (CFCs) from refrigerants, air conditioners, and aerosols
- Halons used in fire extinguishers
- Carbon tetrachloride and methyl chloroform from industrial solvents
- Emission of nitrogen oxides (NOx) from jet aircraft and vehicles
NEET aspirants should remember that these substances release chlorine and bromine atoms which break down ozone molecules in the stratosphere.
3. How does ozone layer depletion affect humans and the environment?
Ozone layer depletion increases the amount of harmful UV radiation reaching Earth's surface, which affects:
- Human health: higher risk of skin cancer, sunburn, cataracts, and immune system suppression
- Plants: reduced crop yields and direct DNA damage
- Wildlife: especially affects marine ecosystems like phytoplankton
- Material degradation: damages plastics, wood, and rubber
These impacts are frequently highlighted in NEET Biology.
4. What is the chemical reaction involved in ozone depletion (NEET syllabus)?
Ozone depletion occurs through chemical reactions involving chlorine and bromine from ODS. The main steps are:
- CFCs reach the stratosphere and are broken down by UV light, releasing chlorine atoms.
- Chlorine reacts with ozone:
Cl + O3 → ClO + O2 - ClO reacts with an oxygen atom:
ClO + O → Cl + O2 - The chlorine atom is re-released, destroying more ozone molecules (catalytic cycle).
This process is fundamental for NEET MCQs and theory questions.
5. What is the ozone hole and where is it most severe?
The ozone hole refers to a significant thinning of the ozone layer, especially over the Antarctic region during spring (August to October). Key points for NEET exams:
- Seasonal and most severe above Antarctica
- Detected by satellite observations since the 1980s
- Caused by accumulation of ODS during polar winter, leading to rapid ozone destruction in spring
6. How can ozone layer depletion be prevented? (NEET important)
Prevention of ozone layer depletion relies on reducing the use of ODS and promoting international agreements. Effective measures include:
- Phasing out CFCs, Halons, and other ODS
- Using eco-friendly refrigerants and aerosols
- Supporting and following global treaties like the Montreal Protocol (1987)
- Raising awareness about ozone safety
NEET students should highlight the significance of such preventive steps in answers.
7. What is the Montreal Protocol and why is it important for ozone protection?
The Montreal Protocol (1987) is an international treaty designed to protect the ozone layer by phasing out ozone-depleting substances (ODS). Importance for NEET and exams:
- Signed by most countries worldwide
- Helped reduce the production and consumption of CFCs, Halons
- Marked as one of the most successful environmental agreements
- Contributed to the gradual recovery of the ozone layer
8. Why are CFCs considered harmful to the ozone layer in NEET Biology?
Chlorofluorocarbons (CFCs) are harmful because they release chlorine atoms in the stratosphere, which directly destroy ozone molecules through a chain reaction. For NEET Biology:
- CFCs have a long atmospheric life and reach the stratosphere intact
- Each chlorine atom can break down thousands of ozone molecules
- Main sources are refrigeration, air conditioning, and spray propellants
9. How does ozone differ from oxygen? (Important for NEET)
Ozone (O3) is a triatomic molecule, while oxygen (O2) is diatomic. Key differences relevant to NEET:
- Ozone acts as an atmospheric shield, absorbing UV radiation
- Oxygen is essential for respiration but does not block UV rays
- Ozone is less stable and has a sharp smell; oxygen is stable and odorless
10. Name two international efforts taken to control ozone layer depletion. (Scraped)
Two major international efforts to control ozone depletion are:
- Montreal Protocol (1987): Phases out the production of key ozone-depleting substances.
- Vienna Convention (1985): Provides a framework for international cooperation to protect the ozone layer.
Both are frequently asked in NEET and school biology exams.
11. What are the harmful effects of UV radiation due to ozone layer depletion? (Scraped)
Depletion of the ozone layer leads to increased UV radiation exposure, which causes:
- Higher risk of skin cancer and sunburns
- Increased cases of eye cataracts
- Weakening of the human immune system
- Reduced crop yields and aquatic life damage
Understanding these effects is crucial for NEET Biology.
12. What steps can students take to help protect the ozone layer? (Scraped)
Students can contribute to ozone layer protection by:
- Avoiding use of products with CFCs or harmful aerosols
- Spreading awareness about ozone layer conservation
- Supporting eco-friendly appliances and green technologies
- Recycling and reducing pollution
Participating in such initiatives is a good example for NEET preparation and social responsibility.