What is Allosteric Enzyme and How Allosteric Regulation Works? Understand Enzyme Kinetics
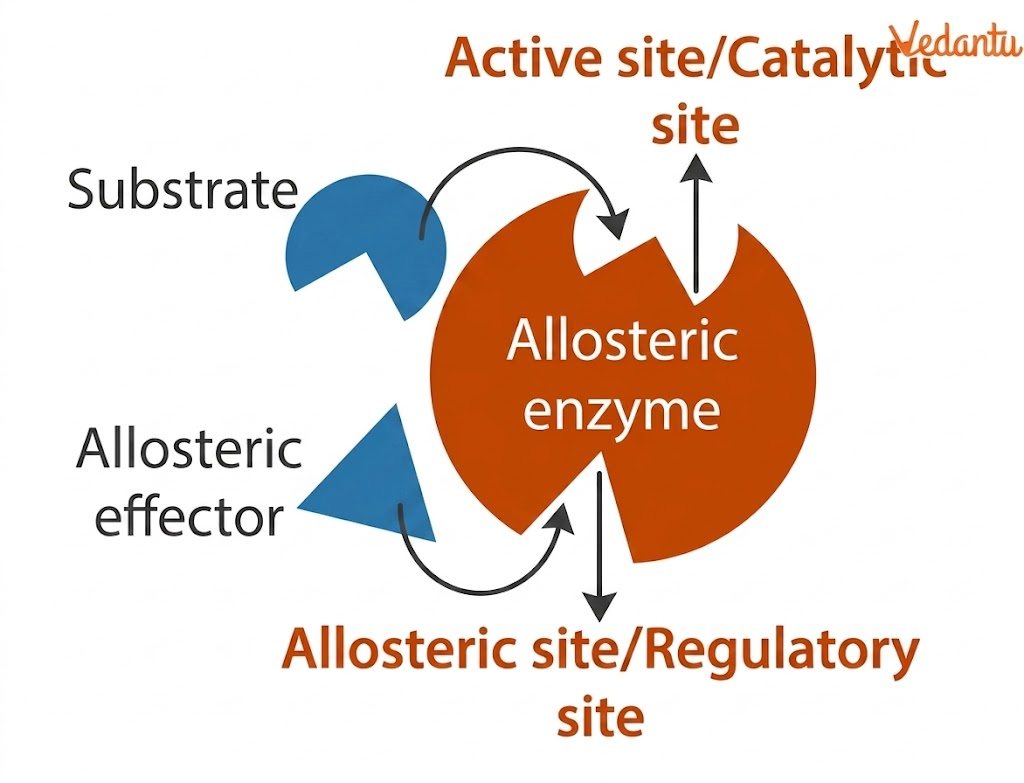
An allosteric enzyme is a regulatory enzyme that contains, in addition to its active site, one or more allosteric sites where specific molecules can bind. These molecules are called effectors or regulators. When an effector binds at the allosteric site, it causes a conformational change in the enzyme. This structural change alters the catalytic activity of the enzyme, either increasing it or decreasing it.
This form of regulation is extremely important in living systems because biochemical pathways cannot be allowed to run randomly. Processes such as metabolism, hormone responses, biosynthesis, and energy production require precise control. Allosteric regulation helps cells respond quickly to internal and external changes without needing to synthesize a new enzyme every time conditions change. That is why allosteric enzymes are considered one of the most important control points in biochemistry.
Simple Definition
An allosteric enzyme is an enzyme that has:
an active site for the substrate
a separate regulatory site for effector molecules
the ability to change its activity through conformational change
Why are Allosteric Enzymes Important?
Allosteric enzymes are important because they regulate the rate of biochemical pathways. Many metabolic pathways contain one rate-limiting step, and this step is often controlled by an allosteric enzyme.
Importance of Allosteric Enzymes
They help maintain metabolic balance.
They prevent overproduction of pathway products.
They allow quick response to changing cellular needs.
They are involved in feedback inhibition and activation.
They coordinate major pathways like carbohydrate, lipid, and nucleotide metabolism.
For example, if a cell already has enough of a certain end product, that product may act as an allosteric inhibitor of an earlier enzyme in the pathway. This prevents wasteful energy expenditure.
Properties of Allosteric Enzymes
Allosteric enzymes have several characteristic features that make them different from ordinary Michaelis-Menten enzymes.
1. Presence of an additional binding site
These enzymes possess one or more binding sites besides the active site. The active site binds the substrate, while the allosteric site binds a regulator or effector molecule.
2. Conformational flexibility
The enzyme can exist in different structural forms. Binding of an effector changes the enzyme’s conformation and affects its catalytic efficiency.
3. Regulation by activators and inhibitors
The effector may be:
an activator, which increases enzyme activity
an inhibitor, which decreases enzyme activity
4. Multiple allosteric sites may be present
An enzyme molecule may contain more than one regulatory site, allowing it to respond to several signals at once.
5. Cooperative behaviour
Many allosteric enzymes show cooperativity, meaning binding of substrate to one subunit affects binding at other subunits.
6. Sigmoidal kinetics
Unlike simple enzymes that show a hyperbolic velocity-substrate curve, allosteric enzymes often show an S-shaped or sigmoidal curve.
These features are highly testable and often asked in conceptual biology and biochemistry questions.
Structure of Allosteric Enzyme
Although the exact structure varies from enzyme to enzyme, many allosteric enzymes are made of multiple subunits. These subunits may include:
catalytic subunits, which contain the active site
regulatory subunits, which bind effector molecules
The provided content describes the substrate-binding part as the C-subunit and the effector-binding part as the R-subunit or regulatory subunit.
This subunit arrangement helps explain why a change in one part of the enzyme can influence the behaviour of the entire protein complex.
Allosteric Enzyme Regulation
Allosteric enzyme regulation is the control of enzyme activity through effector binding at the allosteric site. This regulation can either increase or decrease enzyme activity depending on the type of effector and the metabolic need of the cell.
There are two major ways to classify allosteric regulation:
based on the relationship between substrate and effector
based on whether the effect is activation or inhibition
Homotropic and Heterotropic Allosteric Enzyme Regulation
Homotropic Regulation
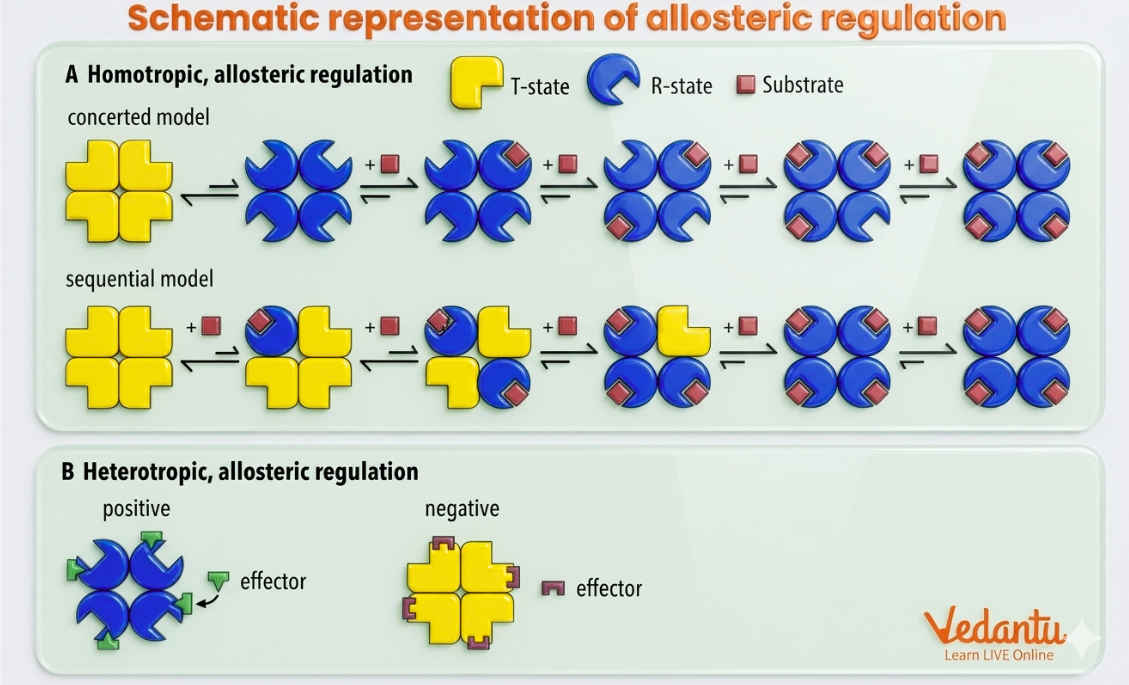
In homotropic regulation, the substrate itself also acts as the effector. In other words, binding of one substrate molecule influences binding of additional substrate molecules. This usually leads to cooperativity and often activation.
Example
The content gives the example of oxygen binding to haemoglobin as a homotropic interaction. Although haemoglobin is not an enzyme, it is a classical example used to explain cooperative binding.
Heterotropic Regulation
In heterotropic regulation, the substrate and the effector are different molecules. The effector may either activate or inhibit the enzyme.
Example
The content mentions carbon dioxide binding to haemoglobin as a heterotropic example. Again, while haemoglobin is not an enzyme, the concept helps explain how one ligand can alter the binding of another.
Key Difference
Homotropic: substrate itself acts as regulator
Heterotropic: a different molecule acts as regulator
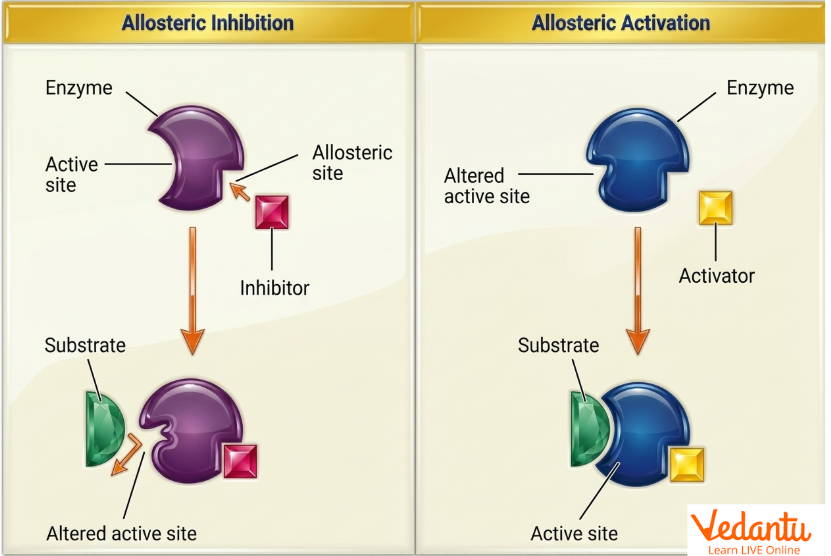
Allosteric Enzyme Inhibition
Allosteric enzyme inhibition occurs when an inhibitor binds to the allosteric site and decreases enzyme activity. This binding changes the shape of the enzyme and reduces the efficiency of substrate binding or catalysis.
What Happens During Allosteric Inhibition?
inhibitor binds at regulatory site
conformational change occurs
active site becomes less favourable
enzyme activity decreases
Allosteric inhibition is often involved in feedback regulation, where the final product of a pathway inhibits an early enzyme in that same pathway.
Why is it useful?
This prevents unnecessary accumulation of products and conserves energy.
Allosteric Enzyme Activation
Allosteric activation occurs when an activator binds to the allosteric site and increases enzyme activity. This changes the enzyme structure in such a way that substrate binding becomes easier or catalytic turnover becomes faster.
What Happens During Activation?
activator binds at regulatory site
conformation shifts to more active state
substrate affinity increases
catalytic efficiency improves
This is especially useful when a metabolic pathway needs to be accelerated quickly.
Allosteric Enzyme and Their Kinetics
The kinetics of allosteric enzymes are different from ordinary enzymes. Simple enzymes often follow Michaelis-Menten kinetics and show a hyperbolic graph between velocity and substrate concentration. Allosteric enzymes, by contrast, usually show cooperative kinetics and produce a sigmoidal curve.
Why is the Graph Sigmoidal?
At low substrate concentration, activity is low because substrate binding is difficult. Once some substrate molecules bind, the enzyme changes conformation and becomes more favourable for further substrate binding. As a result, activity rises sharply over a small range of substrate concentration.
This produces the characteristic S-shaped graph.
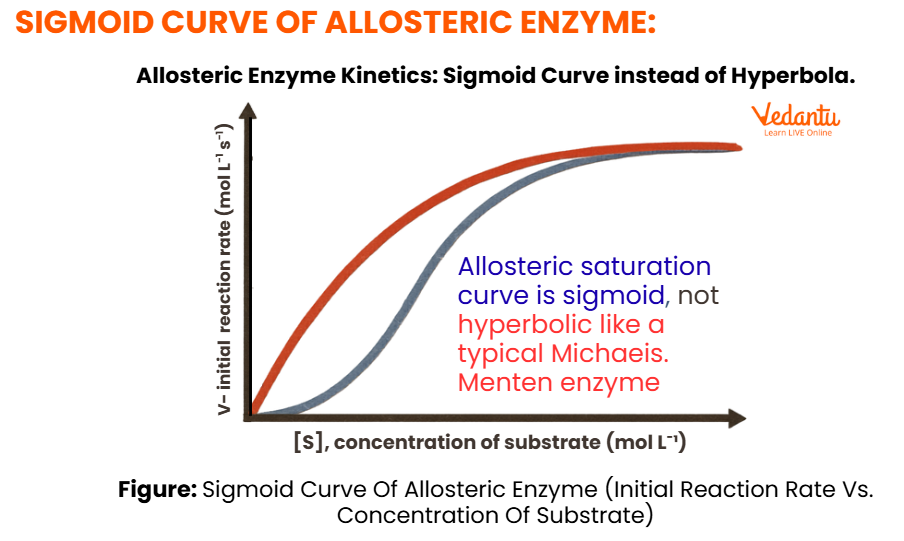
Key point for NEET
Velocity vs substrate concentration graph of an allosteric enzyme is sigmoidal, not hyperbolic.
Allosteric Enzyme Graph
The allosteric enzyme graph is commonly drawn as a plot of reaction velocity versus substrate concentration.
Features of the Allosteric Graph
S-shaped or sigmoidal curve
low activity at low substrate concentration
sharp increase after threshold
eventually reaches saturation
Comparison with Ordinary Enzymes
This graph is one of the easiest ways to identify an allosteric enzyme in exam questions.
Models of Allosteric Enzyme Regulation
Two important models explain the mechanism of allosteric regulation:
Simple Sequential Model
Concerted or Symmetry Model
These models help explain how substrate or effector binding changes enzyme conformation.
Simple Sequential Model of Allosteric Enzyme
The simple sequential model was proposed by Koshland. According to this model, binding of substrate to one subunit induces a conformational change in that subunit from the T state (tensed) to the R state (relaxed). This change then influences neighbouring subunits and promotes similar structural changes.
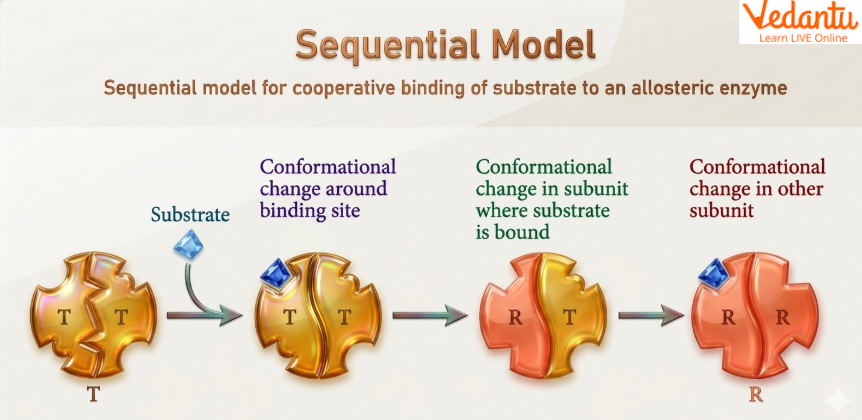
Main Ideas of the Sequential Model
substrate binds according to induced fit theory
one subunit changes first
this influences adjacent subunits
cooperative binding is explained step by step
negative cooperativity can also be explained
The content mentions tyrosyl tRNA synthetase as an example where binding of one substrate inhibits binding of another, illustrating negative cooperativity.
Significance
This model explains how subunits do not need to change together; instead, conformational change can occur progressively.
Concerted Model of Allosteric Enzyme
The concerted model of allosteric enzymes, also called the symmetry model, states that all subunits of an enzyme change conformation simultaneously. The enzyme exists either entirely in the T state (inactive or low affinity) or entirely in the R state (active or high affinity).
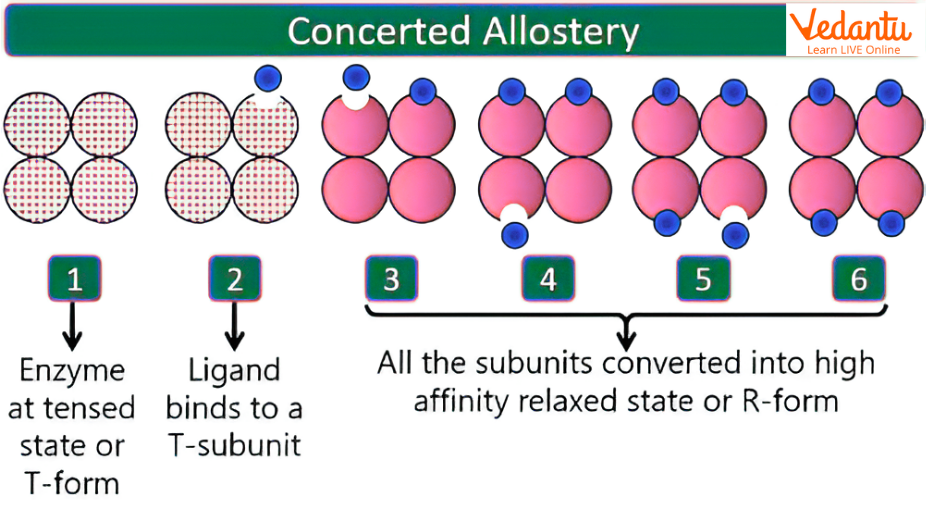
Main Ideas of the Concerted Model
all subunits are in the same state at a given time
transition between T and R is simultaneous
inhibitor shifts equilibrium toward T state
activator shifts equilibrium toward R state
Why is it important?
This model is especially useful in explaining cooperative behaviour where all subunits act in a coordinated manner.
T State and R State in Allosteric Enzymes
Understanding T and R states is essential.
1. T state
Tensed form
lower substrate affinity
relatively inactive
2. R state
Relaxed form
higher substrate affinity
more active
Allosteric regulators work by shifting the equilibrium between these two forms.
Allosteric Enzyme Examples
Several important metabolic enzymes are allosteric. The provided content gives three major allosteric enzyme examples.
1. Aspartate Transcarbamoylase (ATCase)
Aspartate transcarbamoylase is an enzyme involved in pyrimidine biosynthesis. It is a classical example of allosteric regulation and feedback control.
Regulation of ATCase
CTP is the end product of the pathway and inhibits ATCase
ATP activates the enzyme and can overcome CTP inhibition at high concentration
Importance
This ensures balance between purine and pyrimidine nucleotide synthesis. If the purine level is high, pyrimidine synthesis is stimulated to maintain nucleotide balance.
2. Glucokinase
Glucokinase is an important enzyme in glucose homeostasis. It converts glucose into glucose-6-phosphate and promotes glycogen synthesis in the liver. It also helps pancreatic beta cells sense blood glucose concentration for insulin release.
Regulation of glucokinase
The content states that glucokinase has low affinity for glucose, so it becomes especially active when glucose concentration is high. Its activity is regulated by glucokinase regulatory proteins.
Importance
This makes glucokinase suitable for liver function after meals, when blood glucose is elevated.
3. Acetyl-CoA Carboxylase
Acetyl-CoA carboxylase is a major regulatory enzyme of fatty acid synthesis or lipogenesis.
Regulation of acetyl-CoA carboxylase
activated by citrate
inhibited by long-chain acyl-CoA molecules such as palmitoyl-CoA
It is also regulated by phosphorylation and dephosphorylation under hormonal control, including hormones such as glucagon and epinephrine.
Importance
This enzyme links nutritional state, hormonal signals, and lipid metabolism.
Difference Between Ordinary Enzymes and Allosteric Enzymes
This distinction is important in both NEET and higher biochemistry.
Biological Significance of Allostery
Allostery is important because it provides fast, reversible, and sensitive regulation. Unlike gene-level control, which takes more time, allosteric regulation can change enzyme activity immediately in response to metabolite concentration.
Biological Significance
regulates metabolic pathways
prevents product overaccumulation
allows rapid response to cellular need
integrates multiple signals
supports homeostasis
Without allosteric enzymes, metabolic control would be much less efficient.
Quick Revision Points
Allosteric enzymes have a regulatory site besides the active site.
Binding of an effector causes conformational change.
The effector may be an activator or inhibitor.
Allosteric enzymes often show sigmoidal kinetics.
The two main regulatory models are sequential model and concerted model.
Homotropic regulation uses substrate as effector.
Heterotropic regulation uses a different effector.
Important examples include ATCase, glucokinase, and acetyl-CoA carboxylase.
FAQs on Allosteric Enzyme: Regulation, Kinetics, Graph, Models and Examples
1. What is an allosteric enzyme?
An allosteric enzyme is an enzyme that has an additional regulatory site besides the active site. Binding of an effector at this site changes enzyme activity.
2. What is allosteric enzyme regulation?
Allosteric enzyme regulation is the control of enzyme activity by binding of activators or inhibitors at an allosteric site, causing conformational change.
3. Why is the allosteric enzyme graph sigmoidal?
The graph is sigmoidal because many allosteric enzymes show cooperative binding, where substrate binding at one site increases binding at other sites.
4. What is allosteric enzyme inhibition?
It is the decrease in enzyme activity caused by binding of an inhibitor to the allosteric site.
5. What are homotropic and heterotropic allosteric enzymes?
In homotropic regulation, the substrate itself acts as an effector. In heterotropic regulation, the effector is different from the substrate.
6. What is the concerted model of allosteric enzymes?
The concerted model states that all enzyme subunits shift together between T and R states.
7. Give an allosteric enzyme example.
Examples include aspartate transcarbamoylase, glucokinase, and acetyl-CoA carboxylase.