Volume-Temperature graph at atmospheric pressure for a monoatomic gas ( V in m$^3$, T in $^\circ$C) is_______
(A).
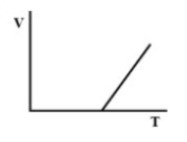
(B).
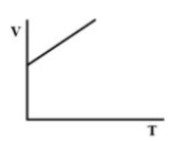
(C).
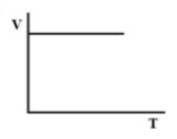
(D).
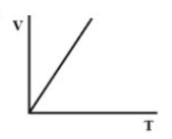




Answer
607.5k+ views
Hint: We all know that a gas follows the ideal gas equation at low pressure and high temperature. One of the parameters can be varied concerning the other parameters by keeping all extra parameters as constant. We can then analyze the variation of pressure, temperature, and volume to each other.
Complete step by step solution:
We know about the ideal gas equation that,
$PV = nRT$
Here, P is the absolute pressure of the gas, V is the volume occupied by the gas, R is the universal gas constant, and T is the absolute temperature, n is the number of moles of a gas.
For deriving this equation, the gas is assumed to be at low pressure and high temperature. Suppose if the pressure of the gas is held constant, then the relationship becomes linear between volume and temperature as R and n along with P is steady now. We get the relation as,
$V = \dfrac{{nR}}{P}T$
Since $\dfrac{{nR}}{P} = C$ it is constant, so the above relation becomes,
$V = CT$
Here we can express the relation as,
$V \propto T$
Here, V is having a linear relation with T. So, there is a straight-line relation between V and T.
Therefore, there must be a straight-line relation between V and T, and the correct option is (D).
Note: Also, we can analyze that when the volume is held constant, the pressure can be varied to temperature and follows a linear relation with temperature. Here the pressure that we are using is absolute and not gauge pressure.
Complete step by step solution:
We know about the ideal gas equation that,
$PV = nRT$
Here, P is the absolute pressure of the gas, V is the volume occupied by the gas, R is the universal gas constant, and T is the absolute temperature, n is the number of moles of a gas.
For deriving this equation, the gas is assumed to be at low pressure and high temperature. Suppose if the pressure of the gas is held constant, then the relationship becomes linear between volume and temperature as R and n along with P is steady now. We get the relation as,
$V = \dfrac{{nR}}{P}T$
Since $\dfrac{{nR}}{P} = C$ it is constant, so the above relation becomes,
$V = CT$
Here we can express the relation as,
$V \propto T$
Here, V is having a linear relation with T. So, there is a straight-line relation between V and T.
Therefore, there must be a straight-line relation between V and T, and the correct option is (D).
Note: Also, we can analyze that when the volume is held constant, the pressure can be varied to temperature and follows a linear relation with temperature. Here the pressure that we are using is absolute and not gauge pressure.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
State and prove Bernoullis theorem class 11 physics CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Which of the following is the hottest month AMay BJune class 11 social science CBSE

What is 1s 2s 2p 3s 3p class 11 chemistry CBSE

Draw the molecular orbital diagram of N2N2 + N2 Write class 11 chemistry CBSE

Which gland is known as mixed gland class 11 biology CBSE




